
Sep 19, 2014 Abstract. Blood transfusion reactions are rare and vary in the degree of harm they cause the patient. In some cases, it may be clear on visual examination that a patient is having a severe reaction to a blood component; however, it may not be clear in all. A blood transfusion is a way of adding blood to your body after an illness or injury. If your body is missing one or more of the components that make up healthy blood, a transfusion can help supply what your body is missing. Depending on how much blood you need, a transfusion can take between 1 and 4 hours.
Almost five million Americans undergo this procedure every year.
A blood transfusion is a procedure during which donated blood is added to your own.
Blood transfusions boost blood levels that are low.
You may need one due to surgery, bleeding, injury, cancer, infection, a blood disorder, a liver problem, or another type of health concern.
Blood contains components such as red blood cells, white blood cells, plasma, and platelets.
A blood transfusion may provide you with whole blood, or with the parts of blood that are most helpful to you.
Sometimes a blood transfusion is performed with your own blood that you've donated ahead of time.
The blood given in a transfusion must be compatible with your blood type. Your blood will be tested to determine if it's A, B, AB, or O, and if it's Rh positive or Rh negative.
Blood banks carefully screen donated blood to identify blood type.
They also test for infectious diseases such as human immunodeficiency virus (HIV), which can be transmitted during a transfusion.
According to the National Heart, Lung, and Blood Institute, almost five million Americans need a blood transfusion each year.
Blood Transfusion Procedure
Blood transfusions are typically performed in a hospital, doctor's office, or other medical facility.
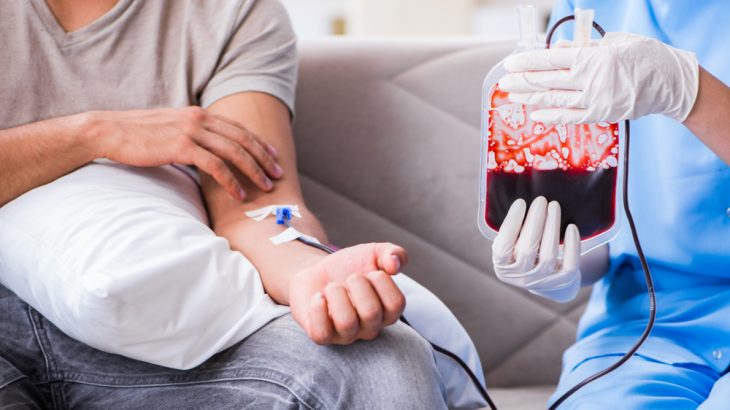
During a blood transfusion, an intravenous (IV) line is placed in a vein in your arm.
Blood or blood components that are stored in a plastic bag are delivered through the IV into your bloodstream.
The procedure typically takes one to four hours.
Tell your doctor or nurse immediately if you experience any of the following symptoms during your transfusion:
- Shortness of breath
- Fever or chills
- Pain at the infusion site
- Unusual itching
- A sense of uneasiness
Before a Blood Transfusion
You won't need to change your diet or limit any activities before receiving a blood transfusion.
Let your doctor know if you've had a transfusion in the past that caused an adverse reaction.
After a Blood Transfusion
After the procedure, you may develop bruising where the needle was inserted.
You may need more blood testing to check how your body is responding to the transfusion.
Blood Transfusion Risks
Blood transfusions are considered safe, but certain complications can happen during or after the procedure. These include:
- Allergic reaction (anaphylaxis)
- Fever
- Lung injury
- Acute immune hemolytic reaction (occurs when the body suddenly attacks donor red blood cells)
- Delayed hemolytic reaction (occurs when the body gradually attacks donor red blood cells)
- Bloodborne infections such as HIV, hepatitis B, or hepatitis C
- Iron overload (too much iron in the blood)
- Graft-versus-host disease (occurs when donor white blood cells attack the recipient's bone marrow)
Blood Products for Special Patient PopulationsWashed red cells may be beneficial for patients with thalassaemia who have repeated severe allergic transfusion reactions or for patients with immunoglobulin A (IgA) deficiency, in which the recipient’s pre-formed antibody to IgA may result in an anaphylactic reaction. Washing of the donor product removes plasma proteins that constitute the target of antibodies in the recipient. Washing may be accomplished using manual or automated techniques. Washed red cells that are not suspended in storage solution must be transfused within 24 hours, and this shorter shelf-life creates the possibility of wastage if patients are not available for transfusion at the time the blood is prepared. Suspension in SAGM after washing allows for shelf life as long as 14 days if a closed circuit is used.Washing usually does not result in adequate leucocyte reduction and should not be used as a substitute for leucoreduction. Instead, washing should be used in conjunction with filtration. In addition, washing of red cell units removes some erythrocytes from the transfusion product, and it is therefore valuable to monitor post-transfusion haemoglobin levels to ensure attainment of the targeted haemoglobin level.Cryopreserved (frozen) red cells is the component derived from whole blood in which red cells are frozen, preferably within 7 days of collection and using a cryopreservant such as glycerol, and then stored at -60°C to -80°C.
This product is used to maintain a supply of rare donor units for patients who have unusual red cell antibodies or who are missing common red cell antigens. Their shelf life of 1-7 days depends on whether they were washed in an open or closed system and whether they were resuspended in SAGM. The shorter shelf life again creates the possibility of wastage.
Approximately 20% of the donor cells are lost in the washing after the freezing process. There is no good evidence about how long these can be stored though in many centres they are kept for 10 years.Red cells obtained by donor apheresis refers to the collection of two units of red cells from the same donor for transfusion of one patient. The reduction of donor exposures may decrease the risk of transmission of infections and developing alloimmunisation and other transfusion-related complications.
This approach creates significant logistical problems as the donors need higher haematocrits, can attend less regularly for donation and the collections are performed using more invasive apheresis techniques. In addition, the collection of two separate bags may create an organisational challenge in ensuring that both units go to the same donor.Neocyte transfusions may modestly reduce blood requirements by using only the younger fraction of red cells form the donor units (Spanos ). However, patients are exposed to a higher number of donors, with a consequent increase in cost, risk of transmission of infections, and risk of developing alloantibodies. Storage time for anticoagulant-preservative solutions with and without additive solution.The maximum duration of storage, as noted on each unit varies with the type of preparation.
However, all of the storage solutions should achieve a mean 24-hour post-transfusion survival of no less than 75% of the transfused red cells. The actual half-life of donor red cells after transfusion is not routinely tested for different additives and for different lengths of storage.The haemoglobin oxygen release function which is extremely important in thalassaemia major is impaired during normal storage due to progressive loss of 2, 3-biphosphoglycerate (2, 3-BPG, previously known as 2, 3- diphosphoglycerate, DPG).
However, the rapid repletion of 2,3-BPG after transfusion generally compensates for the loss of function during storage.Taking all of these issues into consideration and especially in view of the fact that in thalassaemia major decreased recovery and a shortened red cell half-life may increase transfusion requirements and as a consequence the rate of transfusional iron loading, the current practice is to use red cells stored in additive solutions for less than two weeks. In patients with cardiac disease and in small children, particular attention should be paid to the increased volume resulting from additive solutions. In general, for all patients, the lower haematocrit of red cell units containing newer additive solutions should be taken into consideration when calculating the annual rate of transfusional iron loading (see below). Compatibility TestingDevelopment of one or more specific red cell antibodies (alloimmunisation) is an important complication of chronic transfusion therapy (Thompson, Singer, Spanos ). However, the prevalence of alloantibodies varies widely among centers and may be related to the homogeneity of the population, strategies for antigen matching and other factors. It is important to monitor patients carefully for the development of new antibodies and to eliminate donors with the corresponding antigens.
Anti-E, anti-C and anti-Kell alloantibodies are the most common. However, 5-10% of patients develop alloantibodies against other erythrocyte antigens or develop warm or cold antibodies of unidentified specificity.It is recommended that:. All patients with thalassaemia should be transfused with ABO and Rh(C, c, D, E, e) and Kell compatible blood in order to avoid alloimmunisation against these antigens.Some centres use even more extended antigen matching, including full Rh matching, or focus on specific antigens that are more likely to cause alloimmunization in a particular population (Cheng 2012).Most blood banks currently perform a screen for new antibodies and a full crossmatch before each transfusion.
In some centres, blood banks use a newer approach in which the initial approach is an antibody screen only. This alternative approach to formal crossmatch, often referred to as an electronic crossmatch, is only appropriate in blood banks that adhere to strict regulations regarding computer systems, sample labelling and other critical issues. Using either approach, new antibodies must be identified so that blood lacking the corresponding antigen(s) can be used if needed. Not all antibodies are clinically significant and may not be able to destroy apparently incompatible red cells at body temperature. Therefore blood does not need to be antibody negative if the antibody is not reactive at certain temperatures and the blood is crossmatch compatible e.g. Anti N or if they are non significant e.g., CL1 related antibodies.A complete and detailed record of antigen typing, red cell antibodies and transfusion reactions should be maintained for each patient, and should be readily available if the patient is transfused at a different centre. Transfusion of blood from first-degree relatives should be avoided because of the risk of developing antibodies that might adversely affect the outcome of a later stem cell transplant and the risks of transfusion associated graft versus host disease.
The length of time between the sample acquisition and antibody screen and the transfusion of blood for regularly transfused patients is usually 72 hours but may be as long as one week in centres with full Rh and Kell antigen matching in patients who are regularly transfused. The primary concern related to the time between the antibody screen and transfusion is the appearance of new and therefore undetected antibodies during this interval.
Transfusion ProgrammesThe recommended treatment for thalassaemia major involves lifelong regular blood transfusions, usually administered every two to five weeks, to maintain the pre-transfusion haemoglobin level above 9-10.5 g/dl. This transfusion regimen promotes normal growth, allows normal physical activities, adequately suppresses bone marrow activity in most patients, and minimises transfusional iron accumulation (Cazzola, Cazzola ). A higher target pre-transfusion haemoglobin level of 11-12 g/dl may be appropriate for patients with heart disease, clinically significant extramedullary haematopoeisis or other medical conditions, and for those patients who do not achieve adequate suppression of bone marrow activity at the lower haemoglobin level. Sometimes back pain occurs prior to blood transfusion and may also respond to a higher pre-transfusion haemoglobin level.
Although shorter intervals between transfusions may reduce overall blood requirements, the choice of interval must take into account other factors such as the patient’s school or work schedule and other lifestyle issues.The schedule outlined above has been shown to minimize iron loading, while suppressing bone marrow expansion in Italian patients with thalassaemia major (Cazzola, Cazzola ). The optimal regime with other transfusion dependent phenotypes such a E-Beta thalassaemia has not been formally studied and may not be the same, as there is some evidence that lower haemoglobin values may be tolerated in patients with E-Beta thalassaemia.
However in the absence of prospective data to show that low transfusion regimes achieve the same outcomes in such patients, the same approach as for other patients is currently recommended.The decision to initiate lifelong transfusion therapy should be based on a definitive diagnosis of thalassaemia. This diagnosis should take into account the molecular defect, the severity of anaemia on repeated measurements, the level of ineffective erythropoiesis, and clinical criteria such as failure to thrive or bone changes. The initiation of regular transfusion therapy for severe thalassaemia genotypes usually occurs in the first two years of life. Some patients with milder forms of thalassaemia who only need sporadic transfusions in the first two decades of life may later need regular transfusions because of a falling haemoglobin level or the development of serious complications. The risk of alloimmunisation appears to be greater in patients who begin transfusion therapy after the first few years of life (Spanos, Michail-Merianou, see ). Presence of alloantibodies and autoantibodies (see below) may severely compromise transfusion therapy in patients with thalassaemia intermedia, for example, who receive their first transfusions in adolescence or later.
Age and alloimmunisation in thalassaemia.Recommendations regarding the volume of transfused red cells are complicated by the use of different anticoagulant-preservatives and additive solutions. For CPD-A units with a haematocrit of approximately 75%, the volume per transfusion is usually 10-15 ml/kg. Units with additive solutions usually have lower haematocrits in the range of 60-70%, and consequently larger volumes are needed to administer the same red cell mass (see ). For most patients, it is usually easier to avoid these differences in red cell concentration by ordering a certain number of units (e.g., one or two) rather than a particular volume of blood. Younger children may require a fraction of a unit to avoid under- or over- transfusion. For such children or for others who may need a specific volume, the following calculation is generally used (Davies Transfusion ).
Guidelines for choosing how much blood to transfuse.(Desired – actual Hb) × weight × 3/haematocrit of transfused unit = ml to be transfusedMost transfusions of 2 or 3 donor units are administered over 3-4 hours. However, an ongoing study in two London thalassaemia centres suggests that in very carefully selected patients free of cardiac disease and not receiving large volumes, transfusions can be administered at the rate of one unit per hour. Patients with cardiac failure or very low initial haemoglobin levels should always receive smaller amounts of red cells and/or have slower rates of infusion.As an example, to raise haemoglobin level by 4 g/dl in a patient weighing 40 kg and receiving AS1 blood with a haematocrit of 60% would require 800 ml. This calculation assumes a blood volume of 70 ml/kg body weight.The post-transfusion haemoglobin should not be greater than 14-15 g/dl as higher post transfusion haemoglobin values risk hyperviscocity and stroke.
Post transfusion haemoglobin should be measured occasionally to assess the rate of fall in the haemoglobin level between transfusions. This decline may be helpful in evaluating the effects of changes in the transfusion regimen, the degree of hypersplenism, or unexplained changes in response to transfusion.Thus the currently accepted mean target is 12 g/dl with a post-transfusion haemoglobin of 14-15 g/dl and a pre-transfusion haemoglobin of 9.0-10.5 g/dl.
Transfusion and the SpleenThe transfusion requirements in unsplenectomised patients are generally higher than splenectomised patients. In a study of thalassaemia major patients who required more than 250 ml of packed red cells/kg/yr, splenectomy decreased the annual iron loading by an average of 39% (Graziano ). More recently, work has shown that average transfusion requirements are about 30% higher is unsplenectomised (0.43 mg/kg/day) than splenectomised thalassaemia major patients (0.33 mg/kg/day) (Cohen ).
With modern chelation regimes, this is seldom a justification for splenectomy unless blood transfusion rates increase into unmanageable ranges, in the context of an enlarging spleen. Broad categorisation of immune-mediated transfusion- reactions and reported frequencies.Nonhaemolytic febrile transfusion reactions were common in past decades, but have been dramatically reduced by leucoreduction, especially pre-storage leucoreduction, which sharply reduces cytokine accumulation and leucocyte alloimmunisation. In the absence of effective leucoreduction, patients experiencing such reactions should be given antipyretics before their transfusions. Since fever may accompany a haemolytic transfusion reaction or the administration of a unit with bacterial contamination, these other causes should always be considered in a patient who develops fever during administration of red cells.Allergic reactions are usually due to plasma proteins and range from mild to severe.
Milder reactions include urticaria, itching and flushing, and they are generally mediated by IgE. More severe reactions, such as stridor, bronchospasm, hypotension or other symptoms of anaphylaxis may occur, especially in patients with IgA deficiency and anti-IgA antibodies.
Occasional mild allergic reactions often can be prevented by the use of antihistamines or corticosteroids before transfusion. Recurrent allergic reactions can be markedly reduced by washing the red cells to remove the plasma. Patients with IgA deficiency and severe allergic reactions may require blood from IgA -deficient donors.Acute haemolytic reactions begin within minutes or sometimes hours of initiating a transfusion and are characterised by the abrupt onset of fever, chills, lower back pain, a sense of impending death, dyspnea, haemoglobinuria and shock. These unusual reactions most commonly arise from errors in patient identification or blood typing and compatibility testing. The risk of receiving the wrong blood is greater for a patient with thalassaemia who travels to another centre or is admitted to a hospital not familiar with his/her case and medical history. Haemolytic reactions in these patients can still be avoided by (1) the use of optimal methods for identifying the patients and labeling of the sample when blood is obtained for crossmatch, (2) proper linkage of the sample to the donor unit in the blood bank, (3) adherence to standard protocols for screening for antibodies and carrying out the necessary full crossmatching of donor units and (4) use of multiple patient identifiers before transfusing the blood. In many transfusion units, two staff members check the identification of the unit and the recipient prior to beginning the transfusion.
If signs and symptoms suggest an acute haemolytic reaction, the transfusion should be stopped immediately and intravenous fluids should be administered to maintain intravascular volume. Diuretics may help to preserve renal function. Disseminated intravascular coagulation (DIC) may require additional measures such as heparin.
The identification of the patient and the donor unit should be re-checked. The blood bank should also be alerted to the possibility of an undetected alloantibody.Alloimmunisation, as described above, is a common complication of transfusion therapy, occurring in as many as 10-20% of patients with thalassaemia. Alloimmunisation is more common in children who begin transfusion therapy after 1-3 years of age than in those who begin transfusion therapy earlier.
Some evidence also suggests that new alloantibodies develop more frequently after splenectomy (Thompson ). The use of extended antigen matched donor blood is effective in reducing the rate of alloimmunization.Delayed transfusion reactions usually occur 5-14 days after transfusion and are characterised by unexpected levels of anaemia, as well as malaise and jaundice. These reactions may be due to an alloantibody that was not detectable at the time of transfusion or to the development of a new antibody. A sample should be sent to the blood bank to investigate the presence of a new antibody and to repeat cross-matching of the last administered unit(s).Autoimmune haemolytic anaemia is a very serious complication of transfusion therapy that usually but not always occurs in patients with alloantibodies (Ameen ).
Even red cells from seemingly compatible units (i.e., those units that do not contain the antigen to which there is a known alloantibody) may demonstrate markedly shortened survival, and the haemoglobin concentration may fall well below the usual pre-transfusion level because of destruction of both the donor’s and the recipient’s red cells. The serologic evaluation by the blood bank usually shows an antibody that reacts with a wide range of test cells and fails to show specificity for a particular antigen. Steroids, immunosuppressive drugs and intravenous immunoglobulin are used for the clinical management of this complication, although they may give little benefit. Some patients have also been treated with rituximab, but the effectiveness of its use in this situation is still not well defined.
Absence of widely accepted or routinely used tests for bacterial, viral and other pathogens (e.g., Yersinia enterocolitica, hepatitis A, toxoplasmosis, malaria and babesiosis).While pathogen inactivation systems for red cell products are under development (Solhein, Pelletier ), these are not yet available in routine practice. In many regions of the developing world in which thalassaemia is most common, continued transmission of hepatitis B, hepatitis C and HIV underscores the importance of promoting the quality of national blood transfusion services, including voluntary blood donations, careful donor selection and donor blood screening, and the consistent use of immunizations such as hepatitis B vaccine.

